What is a Boiler Deaerator? What is Their Function, How Do They Work?


Boiler deaerators commonly are used to remove oxygen and other gases from the water that feeds into boilers that generate steam. Deaerators are useful because they remove the gases that attach to the metallic components of the steam system and cause corrosion by forming oxides, or rust.
Both oxygen and carbon dioxide are responsible for corrosion, which is why the majority of boiler deaerators are capable of removing oxygen to levels that virtually eliminate carbon dioxide.
There are two main types of boiler deaerators:
Before you can understand how a boiler deaerator works, you need to learn more about boiler deaerators themselves. We explore the following topics to help you gain a better understanding of the process a boiler deaerator uses to remove dissolved gas from water to protect the boiler system itself from corroding. Continue reading to learn more about the ways in which water deaerators operate and how to ensure your deaerator is functioning efficiently.
A boiler deaerator is a crucial component in steam boiler systems designed to protect the system from the harmful effects of dissolved gases in the feedwater. These gases—especially oxygen and carbon dioxide—can cause significant corrosion, leading to maintenance issues and reduced system efficiency.
Removes Dissolved Gases:
Heats Feedwater:
Maintains System Integrity:
Why It Matters:
Without a deaerator, dissolved gases would accelerate corrosion, leading to:
A well-functioning deaerator is essential for maintaining high efficiency and reliable performance in industrial steam systems. This system component plays a silent but vital role in keeping boiler operations safe, efficient, and long-lasting.

Boiler deaerators must condition feed water to remove dissolved gases before it can be used in the boiler system and eventually corrode it. In many cases, feed water is cold and contains several dissolved gases, with oxygen leading the pack.
The conditioning of the boiler’s feed water should meet the goals outlined in the ASHRAE Handbook and the ASME Consensus on Operating Practices for the Sampling and Monitoring of Feedwater and Boiler Water Chemistry in Modern Industrial Boilers.
These goals include removing dissolved oxygen by mechanical means to reduce the need to treat water with chemical oxygen scavengers, removing dissolved gas by mechanical means to protect the steam network and prevent corrosion and failure, removing dissolved non-condensable gas to increase steam system efficiency and potential, and heating cold water to protect the boiler from thermos-shock and prolong the boiler’s life.
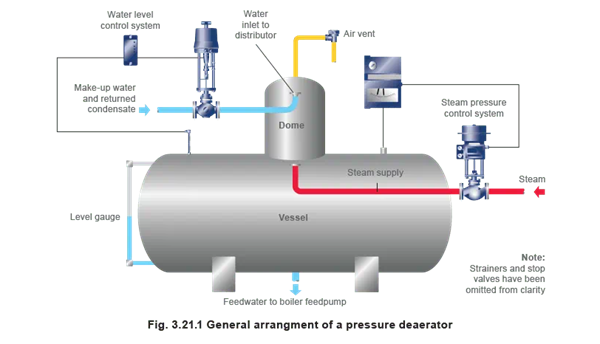
Regardless of the type of boiler deaerator, each functions generally in the same way. When water passes from the feed water tank into the boiler deaerator, it enters through the inlet water connection.
The water flows through a heating and venting section that is filled with steam. The water temperature rises, which releases most of the undissolved gases in it including oxygen and carbon dioxide. When the water flows through the deaerator, it passes to the scrubber section. It is in this section where the last step of deaeration takes place because it scrubs the water with steam that is free of oxygen.
Then, the steam goes through a stainless steel spray valve that breaks down the high-velocity steam into a fine mist. The deaerated water flows over to the storage compartment and is ready for use by the boiler while the gases vent to the atmosphere.
By virtually eliminating the amount of dissolved oxygen and carbon dioxide in the feed water, boiler deaerators help lower operating costs and improve steam quality for facilities.
According to CleanBoiler.org, mechanical deaeration alone cannot ensure complete oxygen removal. Deaerator manufacturers state that properly operating deaerators can reduce dissolved oxygen concentrations in feed water to 0.005 cc per liter, or 7 ppb, and 0 free carbon dioxide. However, plant oxygen levels vary from 3 to 50 ppb, which makes chemical removal of oxygen in feed water with oxygen scavengers necessary.
Best practices with oxygen scavengers include placing them in the deaerator’s storage tank so they have the most time possible to react with remaining dissolved oxygen. However, some conditions warrant placing oxygen scavengers in other locations.
One commonly used oxygen scavenger is sodium sulfite. Some higher pressure boilers, however, cause the sulfite to decompose and enter the steam, which may pose a hazard in condensate systems and condensing steam turbines. It’s worth noting that a change in demand for chemical oxygen scavengers could point to a failing deaerator.

Boiler deaerators feature a mechanical design that enables them to use steam pressure and temperature to condition feed water. When the temperatures, pressure, and dissolved oxygen levels are within design limits, boiler deaerators operate at maximum efficiency.
It is important to check the temperature and pressure daily and log the data into a database to conduct trend analysis. Should any fluctuations be present, you should schedule maintenance for the deaerator controls.
To determine how effectively your boiler deaerator is operating, you should conduct regularly scheduled annual dissolved oxygen studies (DO2 studies) to measure the dissolved oxygen level at the deaerator’s outlet. Physical inspections of deaerators also show whether an internal component failed.
Be prepared to obtain a sample from the deaerator’s storage section and to disconnect the oxygen scavenger system from the deaerator while the feed water sample is being collected.
Boiler deaerators serve a critical role in protecting steam systems from corrosion. It is important to understand how they operate and how to eliminate dissolved oxygen in the deaerator with the assistance of oxygen scavengers in order to determine whether your boiler deaerator is operating efficiently.
Our sales engineers are experts in automatic asset tracking, tagging and identification,a nd can answer all your questions. Get in touch now.
Lets Talk ›Enter your information and get a free guide of the key design considerations applicable to industrial control panel design, including schematics, relevant regulatory standards, and design considerations relating to every facet of effective control panel design for industrial equipment and machinery.